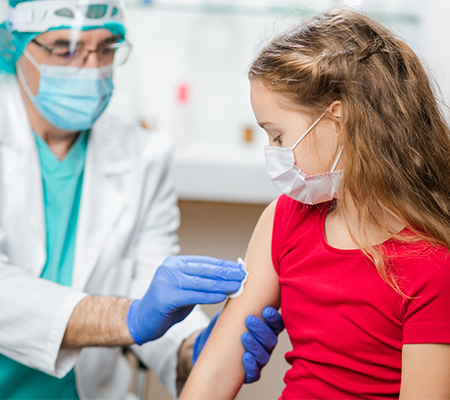
Children 5-11 years of age are now eligible for the pediatric Pfizer-BioNTech COVID-19 vaccine. As a parent, you may have questions about giving your child the vaccine.
Children 5-11 years of age are now eligible for the pediatric Pfizer-BioNTech COVID-19 vaccine. As a parent, you may have questions about giving your child the vaccine.
Dr. Thomas Boyce, pediatric infectious disease doctor and researcher with Marshfield Children’s, understands why parents may have questions.
“For parents, it may be harder to make a decision about vaccinating their children than about getting the vaccine for themselves,” said Dr. Boyce. “However, we now have strong scientific evidence that the Pfizer-BioNTech COVID-19 vaccine is safe and effective. The benefits of vaccination far outweigh the risks.”
Boyce acknowledges that children are hospitalized less frequently than adults for COVID-19. However, as of November 2021 in the U.S., more than 8,000 children have been hospitalized due to COVID-19 and 800 children have died.
About a third of children hospitalized due to COVID-19 do not have any underlying conditions, so even healthy children are at risk for serious COVID-19. Besides hospitalization and death, COVID-19 in children can lead to the severe inflammation of organs and long-term complications.
The best way to prevent serious COVID-19 is to get children ages 5-11 vaccinated against COVID-19. Below are three things to know about the pediatric Pfizer-BioNTech COVID-19 vaccine for children ages 5-11.
Specifics about the shot
The pediatric Pfizer-BioNTech COVID-19 vaccine is given in two doses, 21 days apart as an injection into the muscle. It can be given with other pediatric vaccines, including the flu shot.
“During clinical trials, children experienced fewer side effects than adolescents or young adults. The most common side effects were a sore arm, mild fatigue and headache,” Dr. Boyce said. “Arm swelling, redness and fever are uncommon and occur in less than 1 in 5 children. Side effects are usually mild and resolve within 1-2 days.”
For children between 5-11 years, a smaller dose of the pediatric Pfizer-BioNTech COVID-19 vaccine is given than to people 12 and older. American Academy of Pediatrics recommends that children who are younger than 12 years old for their first injection get the lower dose for both the first and second doses.
Why doctors recommend the vaccine
The pediatric Pfizer-BioNTech COVID-19 vaccine reduced the risk of symptomatic COVID-19 in children by 91% during the phase three clinical trial. About 1,500 children received the vaccine in the clinical trial. The vaccine has also been shown to be highly effective against death and hospitalization due to COVID-19 in all age groups.
Children should still get vaccinated even if they already had COVID-19.
“Natural immunity after COVID-19 infection can provide protection for several months, but the level of protection varies. Antibody levels decline over time and reinfections occur,” Dr. Boyce said. “People with prior infection that have also received a COVID-19 vaccine have greater protection against COVID-19 compared to those with natural immunity alone.”
There is no increased risk of vaccine side effects or complications for people with a prior infection and vaccination.
An added benefit of vaccinating children is to protect those around them. Even children with mild or asymptomatic COVID-19 can spread infection to high-risk contacts, such as grandparents, often with devastating results.
Heart inflammation risk
Serious or long-term health risks are unlikely after receiving the COVID-19 vaccine. However, rare cases of heart inflammation, commonly referred to as myocarditis, have been reported after people received an mRNA COVID-19 vaccine, such as the Pfizer-BioNTech COVID-19 vaccine.
It is most common in males after receiving the second dose. There were no cases among children 5-11 in the clinical trial, but rare myocarditis cases may occur when the vaccine is given to millions of children.
The risk of myocarditis after vaccination is much lower than the risk of myocarditis with COVID-19, and all cases of vaccine myocarditis have been mild with full recovery.
“The risk of myocarditis from COVID-19 in adolescents and young adults is 200-500 per million cases. These cases can be severe. The risk of myocarditis from the mRNA vaccines in that age group is about 20 cases per million second doses. These cases are generally mild. So it is on the order of 10 times higher risk with COVID-19 than from the vaccine. It is actually riskier not to get your child vaccinated than to get them vaccinated.”
The consensus in the medical field is that the benefits of vaccination outweigh the risks based on the current level of COVID-19 activity.
The CDC and FDA continue to monitor the safety of the vaccine. You also can sign up your child for the CDC V-SAFE program using a smartphone after vaccination. V-SAFE collects data on symptoms for one year.
Talk to your child’s doctor if you have any questions about the COVID-19 vaccine.


Thank you for the information.
How many kids in that age group have died ? In the US.
Hi Russ,
Prior to November 2021 in the U.S., more than 8,000 children have been hospitalized due to COVID-19 and 800 children have died. No children have died from the COVID-19 vaccine.
Thank you.